Rx-to-OTC Switch
Are you sure you'd like to remove this alert? You will no longer receive email updates about this topic.

Opill Launch Delivers For Perrigo While Growing Pains Slow Overall First-Quarter Results
Perrigo spares little about Opill’s launch, advertising and outlook in Q1 earnings briefing, but management also makes clear it’s a large firm and some other segments of its global consumer health products business are holding back overall results.

Need Advice About Advisory Committees, US FDA? Try Having Some
FDA’s drug center has all but stopped holding meetings at this point, which begs the question: why do they want advice on how to make them better?

FDA Advisory Committee Reform: Does Listening Session Suggest No Major Near-Term Changes?
FDA seeks feedback at 13 June virtual hearing on three general topics related to advisory committees: panel composition, barriers to service for Special Government Employees, and public perception and understanding of the process.

First Generic Of B+L’s OTC Switch Lumify Approved For Dr. Reddy’s Amid Patent Infringement Battle
Canadian firm received approval from FDA for NDA submitted in May 2023 for Lumify Preservative Free. A month earlier, FDA approved Dr. Reddy's ANDA submitted in July 2021 for an equivalent of Lumify’s formulation, brimonidine tartrate 0.025% ophthalmic solution.

Maxwellia Delivers Two UK Menstrual Health Rx-To-OTC Switches
UK switch specialist Maxwellia delivers two “me too” reclassifications in the area of women's health: Evana Heavy Period Relief and Ultravana Period Pain Relief.

Largest PBMs Eliminate Consumers’ Cost Sharing For Opill On Most Commercial Plans
Decisions by “big three” PBMs suggest they’re taking proactive steps to comply with potential HRSA rulemaking to require most health plans, under Affordable Care Act regulations, to cover Opill and other types of OTC birth control without cost sharing.

Poland's President Blocks Rx-To-OTC Switch Of Emergency Contraceptive Pill
Citing a will to protect children's health, Poland's president has blocked a bill which would have restored OTC access to the morning after pill to those aged 15 and above. In response, the health ministry has drawn up a proposal to allow pharmacists to issue prescriptions for emergency contraception.

Perrigo Partnership Makes Opill Star Player On WNBA's Reproductive Health Advocacy Team
Less than a month into launching sales of first oral contraceptive approved for US OTC sales, Perrigo announces multi-year partnership with WNBA focused on building confidence, strength and health equity for women.

FDA Idea To ‘Encourage Competition For New Drugs’ Spares Cutting OTC Switch Market Exclusivity
“FDA is seeking to encourage competition for new drugs by proposing to amend the Hatch-Waxman 3-year exclusivity provisions to ensure that this exclusivity is limited to situations where the new drug applicant is actually seeking such exclusivity,” according to legislative proposals document.
ENVI: Prescription Requirement Only With Evidence Of Antimicrobial Resistance
The EU Parliament's Environment, Public Health and Food Safety committee proposes a “compromise amendment” to the new pharmaceutical directive that would include only antimicrobials for which there is an “identified risk of antimicrobial resistance” to the prescription requirement.
Why Did Germany’s Switch Committee Reject Perrigo’s OTC Daily Contraceptive Proposal?
Skepticism about the need for widening access to the progestogen-only contraceptive pill, fears of associated thrombosis risk with use and the potential cost barriers were among the many reasons for the unanimous rejection of Perrigo's Rx-to-OTC switch application for desogestrel (75μg) for oral use.

Sweden Seeks To Tackle Opioid Overdose Deaths With Naloxone Rx-To-OTC Switch
Opioid antagonist naloxone will soon be available without prescription in Sweden following a successful Rx-to-OTC application from Norway's DNE Pharma.
Poland's Government Pushing To Restore OTC Access To Morning After Pill
As Poland is ranked the worst country in Europe for contraception access by a group of EU parliamentarians, the country's government has voted through a bill which would make the morning after pill available without a prescription for the first time in more than six years.
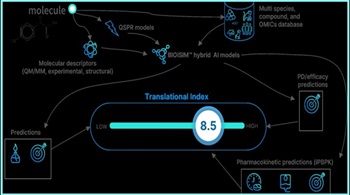
When Using Artificial Intelligence In Pharma R&D, Start With Identifying Problem To Solve
VeriSIM uses generative AI for questions such as changing a drug molecule’s chemistry and machine learning to better predict potential biological implications. “When a child is learning a language, you're teaching a child starting from A-to-Z structures, and then suddenly they start speaking. Then they start learning different things, and then they get into problem solving. That's really what AI is getting really good at now,” says CEO Jo Varshney.

VeriSIM Expects AI Use To Trim Margin For Failure In OTC Switches, Help Boost Revenue Margins
“It's a very expensive process for an OTC drug because you don't make significantly high margins compared to a blockbuster cancer drug. For pharma companies utilizing our platform, you can reduce that efficiency challenge and increase the probability of success,” says VeriSIM CEO Jo Varshney.

Perrigo Ships Opill To Stores, But Contraceptive Access Advocates Say It Doesn’t Deliver On Price
First US OTC daily oral contraceptive shipped to major retailers and pharmacies and will be available on shelves nationwide and online later in March. Perrigo says pre-orders for Opill are available currently from select online retailers.
You must sign in to use this functionality
Authentication.SignIn.HeadSignInHeader
Email Article
All set! This article has been sent to my@email.address.
All fields are required. For multiple recipients, separate email addresses with a semicolon.
Please Note: Only individuals with an active subscription will be able to access the full article. All other readers will be directed to the abstract and would need to subscribe.